 |
 |
 |
|
||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
Applications in Confocal Microscopy
Specimen Preparation Using Synthetic Fluorophores and Immunofluorescence
Confocal microscopy was becoming more than just a novelty in the early 1980s due to the upswing in applications of widefield fluorescence to investigate cellular architecture and function. As immunofluorescence techniques, as well as the staining of subcellular structures using synthetic fluorophores, became widely practiced in the late 1970s, microscopists grew increasingly frustrated with their inability to distinguish or record fine detail in widefield instruments due to interference by fluorescence emission occurring above and below the focal plane. Today, confocal microscopy, when coupled to the application of new advanced synthetic fluorophores, fluorescent proteins, and immunofluorescence reagents, is one of the most sophisticated methods available to probe sub-cellular structure. The protocols described in this section address the specimen preparation techniques using synthetic fluorophores coupled to immunofluorescence that are necessary to investigate fixed adherent cells and tissue cryosections using widefield and confocal fluorescence microscopy.
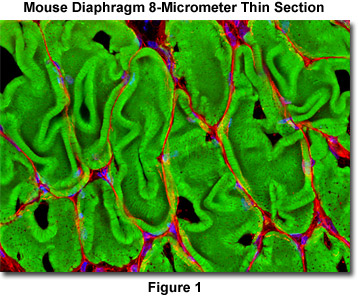
Presented in Figure 1 is a laser scanning confocal image revealing the extensive filamentous actin network present in the smooth muscle tissue of a thin (8-micrometer) cryosection of rat diaphragm. The tissue cryosection was labeled with a cocktail containing Alexa Fluor 488 conjugated to phalloidin (staining actin) and Texas Red-X conjugated to wheat germ agglutinin (targeting lectins). In addition, nuclei in the specimen were counterstained with Hoechst 33342. Images were recorded in grayscale with an Olympus FluoView FV1000 coupled to a BX-81 inverted microscope using Argon-ion (488 nanometer line), violet diode (405 nanometers), and green helium-neon (543 nanometers) lasers. During the processing stage, individual image channels were pseudocolored with RGB values corresponding to each of the fluorophore emission spectral profiles.
Staining Protocols
Triple-Staining Adherent Cells with MitoTracker, Phalloidin (or Phallacidin), and Nuclear Dyes - The vitality and physical properties of adherent cells grown on coverslips in Petri dishes can be readily determined using a popular combination of fluorescent stains that includes one of several MitoTracker probes (for mitochondria) along with phalloidin (or phallacidin) conjugated to common low molecular weight synthetic fluorescent probes. This protocol details a generalized procedure for staining a variety of cell types.
Staining Adherent Cells with Cytokeratin Primary Antibodies and Synthetic Fluorophores - A majority of the common epithelial cell lines derived from humans and laboratory animals produce brightly colored fluorescent specimens detailing the cytokeratin intermediate filament network when stained with a cocktail that includes keratin antibodies conjugated to common low molecular weight synthetic fluorescent probes. In general, the best candidates for cytokeratin immunofluorescence are epithelial cells, such as those derived from kidney. Fibroblasts usually do not respond well to probes for cytokeratin.
Staining Adherent Cells with Intermediate Filament Primary Antibodies and Synthetic Fluorophores - Fibroblast and epithelial cell lines derived from humans and laboratory animals produce brightly colored fluorescent specimens highlighting specific proteins in the intermediate filament network (such as vimentin and desmin) when stained with a cocktail that includes anti-intermediate filament antibodies conjugated to common low molecular weight synthetic fluorescent probes.
Staining Cells and Tissue Cryosections with Tubulin Primary Antibodies, Phallotoxins, and Synthetic Fluorophores - Cell lines and mammalian tissue sections derived from humans and laboratory animals, including intestine, kidney, testes, muscle, liver, and the lungs, produce brightly colored fluorescent specimens detailing the cytoskeletal network when stained with a cocktail that includes tubulin antibodies and phalloidin (or phallacidin) conjugated to common low molecular weight synthetic fluorescent probes. This protocol details a generalized procedure for staining both adherent cells and tissue sections ranging from 5 to 20 micrometers in thickness.
Triple-Staining Tissue Cryosections with Wheat Germ Agglutinin, Phalloidin, and Nuclear Dyes - A majority of the common tissue sections derived from laboratory animals, including intestine, kidney, testes, muscle, liver, and the lungs, produce brightly colored fluorescent specimens detailing a wide variety of anatomical features when stained with a cocktail that includes wheat germ agglutinin and phalloidin (or phallacidin) conjugated to common low molecular weight synthetic fluorescent probes. This protocol details a generalized procedure for staining tissues ranging from 5 to 20 micrometers in thickness.
Immunofluorescence with Brain Tissue Cryosections - Among the antibody targets often labeled in brain tissue sections are glial fibrillary acidic protein (GFAP), neuronal and axonal neurofilaments (both phosphorylated and non-phosphorylated; heavy, medium, and light), class III beta-tubulin, microtubule-associated proteins, blood-brain barrier proteins, synaptic proteins, myelin, and a host of antigens associated with specific diseases.
Immunofluorescence with Brain Tissue Floating Cryosections - Indirect (two-stage) immunofluorescence is a powerful imaging technique that enables the visualization of highly specific targets using a combination of primary (direct) and secondary (indirect) antibodies. Antigenic determinants of interest in brain tissue are first treated with a monoclonal or polyclonal primary antibody from a common host (mouse, rabbit, chicken, rat, donkey, etc.) followed by a secondary antibody directed to the host primary and conjugated to a synthetic or natural fluorophore. This protocol details a generalized procedure for staining frozen brain floating cryosections greater than 30 micrometers in thickness.
Contributing Authors
Nathan S. Claxton, Gregory K. Ottenberg, John D. Griffin, Scott G. Olenych, and Michael W. Davidson - National High Magnetic Field Laboratory, 1800 East Paul Dirac Dr., The Florida State University, Tallahassee, Florida, 32310.
