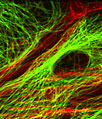
|
 |
 |
|
||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
The FluoViewTM series of laser scanning confocal microscopes offers a choice of several system configurations that are able to meet the needs of an individual researcher or a number of researchers with a variety of different applications. All system configurations and options are compatible with the entire Olympus range of research microscopes offering high-resolution confocal sectioning with the ability to conduct quantitative time-lapse experiments.
Olympus FV1000 - A new generation imaging system for living cells, the FV1000 offers a host of advanced features that enable state-of-the-art research projects to be conducted with ease. The system incorporates two laser scanners in a single compact design for simultaneous excitation and observation. The FV1000 delivers all of the key performance functions required of a confocal laser scanning microscope, minimizing specimen damage following rapid changes in living systems and accurately capturing a full range of information.
Olympus FV300 - The FV300 is a point-scanning, point-detection, confocal laser scanning microscope designed for biology research applications. Excellent resolution, efficiency of excitation, intuitive user interface and affordability are key characteristics of this instrument. When configured with visible-excitation, the microscope permits simultaneous collection of up to 3 detection channels. Maximum optical efficiency is combined with easy, one-touch selection of pinholes and filters.
Olympus FV500 - Discontinued with the introduction of the FV1000, the older FV500 is a fully automated confocal system that can be configured with ultraviolet through infrared lasers and permits simultaneous collection of up to 5 detection channels. Existing FV500 hardware is capable of being matched to either the IX2 inverted research microscope platform or the BX2 upright research microscope platform. Information in this section is provided for users who continue to utilize the FV500 in laboratory applications.
FluoViewTM Confocal Microscope Features
Features and Benefits - Confocal microscopy can improve conventional fluorescence images by recording fluorescence generated from the focal plane within the sample, while rejecting all other light coming from above or below the focal plane. The efficient point-scan/pinhole-detection confocal optics of the FluoViewTM systems virtually eliminate out of focus light to produce high-contrast images with superb resolution. The features listed in this section are available on FV300, FV500, and FV1000 microscope system configurations.
Software Graphical User Interface - The FluoViewTM systems are fully integrated workstations that incorporate user-friendly image acquisition and image analysis software with high-resolution confocal optics that require no user alignment. An intuitive, Windows-based graphic user interface (illustrated below) allows new users to quickly generate images in a wide variety of scanning modes. Standard image formats, including the popular TIFF and AVI algorithms, permit easy, direct export of FluoViewTM images to off-line analysis packages.
Scanning Techniques - The Olympus FluoViewTM confocal microscopes provide innovative scanning techniques for improved performance. Included in the palette are point, line, free line and rectangle scanning modes that make the FV300 or FV500 especially suitable for many time-lapse applications. Sequential scanning can also be performed in order to minimize crosstalk between fluorophore channels by exciting each dye individually, and then assembling the images into a final montage.
Illumination System Components - The FluoViewTM confocal microscopes can be equipped with a variety of laser systems in several different configurations. Among the lasers available for this instrument are the traditional argon-ion and helium-neon, as well as argon-krypton, helium-cadmium, and blue diode lasers. Olympus confocal microscopes can also be adapted to tungsten-halogen and arc-discharge lamps for simultaneous imaging in fluorescence, confocal, and brightfield enhanced contrast techniques (including differential interference contrast). The transmitted light images, while non-confocal, are very useful when superimposed to the confocal image.
Objectives - Olympus continues to pursue the development of advanced optical systems designed to meet the increasing sophistication of demanding applications in optical microscopy. Nothing exemplifies that commitment more clearly than the UIS infinity optics, now established as one of the most successful developments in this field. Chromatic aberration and field curvature are both fully compensated by the objective itself, providing consistently excellent clarity in every method of observation. The UIS system is also valued for its versatility, enabling individual items of equipment to be flexibly combined to meet the particular demands of different needs and applications. These include specialized objectives designed for superior performance in laser scanning applications covering the ultraviolet, visible, and infrared spectral regions.
Acousto-Optic Tunable Filters (AOTFs) - The acousto-optic tunable filter (AOTF) is a specially designed element that controls light intensity by directing acoustic waves through a dense crystalline matrix. The principal feature of the AOTF is the high speed control of laser light intensity and wavelength. The ultrasonic wave introduced into the AOTF crystal forms a compression wave and acts as a diffraction grating due to the re-distribution of the refractive index within the crystal. The grating separates incident laser light into zeroth order and first order diffraction waves. The first order diffraction wave is used, and its wavelength and intensity are controlled by the frequency and intensity of the ultrasonic wave. The AOTF system employed in the FluoViewTM uses multiple acoustic frequencies to control each laser wavelength independently.
FluoViewTM Brochures - For detailed information about the Olympus FluoViewTM and available support equipment for the innovative microscopist, the appropriate brochures may be downloaded. Extensive product photographs, charts, descriptions, sample images, technical specifications, and application notes can help you decide upon the perfect microscope, imaging system, software, and accessories to meet your specific research needs. Included in this section are brochures on research microscopes, digital camera systems, laser systems, objectives, dichromatic mirror units, software, and research microscopes.
FluoViewTM Applications
Three-Dimensional Imaging - The Olympus FluoViewTM microscope software is capable of generating composite and multi-dimensional views of optical section data acquired from z-series image stacks. The three-dimensional rendering features of the software package can be employed to create either a single three-dimensional representation of the specimen or a video (movie) sequence compiled from different views of the specimen volume. These sequences often mimic the effect of rotation or similar spatial transformation that enhances the appreciation of the specimen's three-dimensional character. In addition, the software enables investigators to conduct measurements of length, volume, and depth, and specific parameters of the images, such as opacity, can be interactively altered to reveal internal structures of interest at differing levels within the specimen.
Time Course Experiments - The advanced FluoViewTM confocal software makes possible multi-dimensional views of living cells and tissues that include image information in the x, y, and z dimensions as a function of time (t) and presented in multiple colors (using two or more fluorophores). After volume processing of individual image stacks, the resulting data can be displayed as three-dimensional multicolor video sequences in real time. Note that unlike conventional widefield microscopy, all fluorochromes in multiply labeled specimens appear in register using the FluoViewTM confocal microscope. Temporal data can be collected either from time-lapse experiments conducted over extended periods or through real time image acquisition in smaller frames for short periods of time. The potential for using multi-dimensional confocal microscopy as a powerful tool in cellular biology is continuing to grow as new laser systems are developed to limit cell damage and computer processing speeds and storage capacity improves.
Fluorescence Resonance Energy Transfer (FRET) - The helium-cadmium (HeCd) laser system option for the Olympus FluoViewTM confocal microscopes can provide a useful avenue for resonance energy transfer experiments using the popular cyan and yellow fluorescent protein (CFP and YFP) combination. The 442-nanometer spectral line from the helium-cadmium laser excites CFP very efficiently with virtually no excitation of the YFP fluorophore at the same wavelength. In addition, the Olympus high-performance objectives designed specifically for laser scanning confocal microscopy, PLAPO40xWLSM and PLAPO60xWLSM, are precisely corrected in this wavelength region, and ensure the highest measuring reliability.
PAPP for FRAP Application - The Olympus FluoViewTM programmable acquisition protocol processor (PAPP) software provides easy, reliable flow of experiments in fluorescence recovery after photobleaching (FRAP). Translational mobility (lateral diffusion coefficients) of fluorescently labeled macromolecules and small fluorophores can be readily determined by photobleaching recovery techniques coupled to laser scanning confocal microscopy. In FRAP, a very small, selected region (several micrometers in diameter) is subjected to intense illumination, usually with a laser, to produce complete photobleaching of fluorophores in the region. The result is a dramatic reduction or annihilation of fluorescence. After the photobleaching pulse, the rate and extent of fluorescence intensity recovery in the bleached region is monitored as a function of time to generate information about repopulation by fluorophores and the kinetics of recovery.
Specialized Confocal Techniques - Olympus continues to pursue the development of advanced accessories designed to meet the increasing sophistication of demanding applications that expand the possibilities of confocal microscopy. From slice patching systems and high numerical aperture water immersion objectives for live-cell imaging to specialized software for ratio imaging and time-lapse analysis, the ongoing development of FluoViewTM laser systems, scanning units, and software packages remains a top priority for Olympus scientists and engineers.