|
 |
 |
|
||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
Confocal Microscopy Image Gallery
Opossum Kidney Epithelial Cells (OK Line)
The OK cell line was derived from the kidney of an adult female North American opossum and was originally intended for use as a source of X chromosomes for studies of X inactivation. The line was soon discovered, however, to display many characteristics of kidney proximal tubular epithelial cells and has since been commonly utilized as a cell culture model for the cell type.

OK cells exhibit a stable nondiploid chromosomal modal number of 23 and display a variety of receptors in culture, including alpha 2 adrenergic, serotonin, parathyroid hormone (PTH), and atrial natriuretic peptide (ANP) receptors. Many studies utilizing OK cells focus upon these receptors, some of which are involved in such notable activities as insulin metabolism and the hormonal regulation of phosphate transport and uptake.
The key unit that comprises the vertebrate kidney is the nephron, which essentially consists of a long tubule and a bundle of capillaries called the glomerulus. There is only a single tubule in each nephron, but there are many different regions of the structure. The proximal tubule is the portion of the tubule nearest Bowmans capsule, which is a sac-like swelling located at the blind end of the tubule that encases the glomerulus. Fluids from the blood contained in the glomerulus are collected in Bowmans capsule, and the filtrate is then passed on to the proximal tubule followed by the other portions of the nephron, including the loop of Henle and the distal tubule, before emptying into a collecting duct connected to the renal pelvis. Within the proximal tubule, the volume and composition of the filtrate is significantly altered, as some substances, such as salts and water, are reabsorbed, and others, including hydrogen ions and ammonia, are secreted.
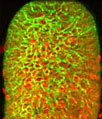
The opossum kidney cells presented in the digital image above were resident in a culture that was immunofluorescently labeled with anti-tubulin mouse monoclonal primary antibodies followed by goat anti-mouse Fab fragments conjugated to Alexa Fluor 568 (pseudocolored green). F-actin in the cells was targeted with Alexa Fluor 488 conjugated to phalloidin (pseudocolored blue), and DNA was counterstained with DRAQ5. In the dividing cell in the center of the image, the DNA can be seen organized into sets of chromosomes, whereas the nucleic acid in the cell along the lower left-hand side is more diffuse, so that the entire nucleus appears red. Images were recorded with a 60x oil immersion objective using a zoom factor of 3.5 and sequential scanning with the 488-nanometer spectral line of an argon-ion laser, the 543-nanometer line from a green helium-neon laser, and the 633-nanometer line of a red helium-neon laser. During the processing stage, individual image channels were pseudocolored with RGB values corresponding to each of the fluorophore emission spectral profiles unless otherwise noted above.
Additional Confocal Images of Opossum Kidney Epithelial (OK) Cells
Imaging F-Actin and Nuclear DNA in Monolayer OK Cell Cultures - A log phase monolayer culture of opossum kidney epithelial cells was stained for the filamentous actin network with Alexa Fluor 488 conjugated to phalloidin. In addition, cell nuclei were targeted with DRAQ5, a far-red fluorescent DNA probe.
Visualizing Mitosis in OK Cell Cultures with Immunofluorescence - The OK cell appearing in this section is in the early anaphase stage of mitosis, the chromosomes appearing to have just become completely separated. The chromosomes were visualized by treating the culture that this cell was resident in with mouse anti-histone (pan) monoclonal antibodies, followed by secondary goat anti-mouse antibodies conjugated to Texas Red.
Opossum Kidney Cells with Alexa Fluor 488, MitoTracker Red CMXRos, and TO-PRO-3 - The adherent culture of opossum kidney (OK) cells presented in this section was labeled for F-actin and the mitochondrial network with Alexa Fluor 488 conjugated to phalloidin and MitoTracker Red CMXRos, respectively. The cells were also probed for DNA with the red-absorbing dye TO-PRO-3.
Distribution of Actin and Microtubules in OK Cells - Immunofluorescence with mouse anti-alpha-tubulin was employed to visualize distribution of the microtubule network in a log phase monolayer culture of opossum kidney cells. The secondary antibody (goat anti-mouse IgG) was conjugated to Alexa Fluor 546 and mixed with Alexa Fluor 488 conjugated to phalloidin to simultaneously image tubulin and the actin cytoskeleton.
Contributing Authors
Nathan S. Claxton, Shannon H. Neaves, and Michael W. Davidson - National High Magnetic Field Laboratory, 1800 East Paul Dirac Dr., The Florida State University, Tallahassee, Florida, 32310.