|
 |
 |
|
||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
 | ||||||||||||||||||||||||
Confocal Microscopy Image Gallery
Rat Brain Tissue Sections
Blood Vessels
The vasculature of the central nervous system is responsible for providing oxygen and glucose to brain, as well as removing lactic acid and other metabolites from the organ. Any disruption of the flow of blood through the brain can have severe consequences.
Transient interference in brain function transpires when ischemia occurs even for only a few seconds, and disturbances in brain circulation that last for several minutes can cause permanent damage. Sudden abnormalities in brain activity related to occlusion or hemorrhaging of a blood vessel are known as strokes. Signs of a stroke vary, but often include the abrupt onset of weakness or numbness in the face or the limbs on the left or right side of the body, loss of vision, acute headache, difficulty producing or understanding speech, physical instability, and dizziness. Paralysis, coma, or death may ensue after a relatively short time so it is extremely important for individuals suffering from a stroke to receive prompt treatment, which frequently involves the administration of anticoagulants or surgery.
Blood vessels are located on the surface of the brain and deeply seated in its tissues. These vessels enter the skull through the small openings known as foramina. Two primary sets of vessels provide blood to the brain: the right and left internal carotid arteries and vertebral arteries. At the base of the brain, the vertebral arteries are joined together into the basilar artery, which connects to the carotid arteries in a vascular loop called the circle of Willis. The arrangement of the circle of Willis allows collateral circulation, so that damage to any one of its components does not necessarily hinder the supply of blood to the tissues of the brain via the anterior, middle, and posterior cerebral arteries. The circle of Willis is not functional in all people, however, since the structure is incompletely formed in a significant segment of the population.
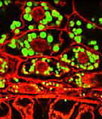
A horizontal thin section of rat brain tissue featuring a blood vessel (shown above) was immunofluorescently labeled for microtubule associated protein 2, which is expressed only in the dendrites and perikarya of neurons, with chicken anti-MAP2 polyclonal antibodies followed by goat anti-chicken secondary antibodies conjugated to Alexa Fluor 488 (yielding green fluorescence emission). Astroglia and intermediate filaments were also targeted in the specimen with rabbit anti-GFAP and mouse anti-vimentin antibodies visualized with goat anti-rabbit and anti-mouse secondaries conjugated to Alexa Fluor 568 (red fluorescence) and Alexa Fluor 647 (pseudocolored magenta), respectively. Cell nuclei were counterstained with Hoechst 33342. Images were recorded with a 20x objective using a zoom factor of 1.6 and sequential scanning with the 405-nanometer spectral line of a blue diode laser, the 488-nanometer line of an argon-ion laser, the 543-nanometer line from a green helium-neon laser, and the 633-nanometer line of a red helium-neon laser. During the processing stage, individual image channels were pseudocolored with RGB values corresponding to each of the fluorophore emission spectral profiles unless otherwise noted above.
Additional Confocal Images of Rat Brain Blood Vessel Tissue Sections
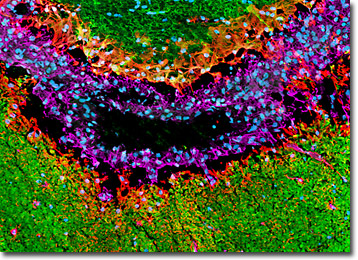
Distribution of MAP2, Vimentin, and GFAP in a Rat Brain Horizontal Tissue Section with Blood Vessel - In a triple immunofluorescence experiment, the horizontal thin section of rat brain presented in this confocal image was fixed, permeabilized, blocked with 10-percent normal goat serum, and then treated with a cocktail of mouse anti-vimentin (intermediate filaments), rabbit anti-GFAP (astroglia), and chicken anti-MAP2 (neuronal dendrites and perikarya) primary antibodies followed by goat anti-mouse, anti-rabbit, and anti-chicken secondary antibodies (IgG) conjugated to Alexa Fluor 647, Alexa Fluor 568, and Alexa Fluor 488, respectively. Subsequently, cell nuclei were counterstained with Hoechst 33342, a nucleic acid probe that fluoresces blue when bound to dsDNA.
Contributing Authors
Nathan S. Claxton, Shannon H. Neaves, and Michael W. Davidson - National High Magnetic Field Laboratory, 1800 East Paul Dirac Dr., The Florida State University, Tallahassee, Florida, 32310.