Confocal Microscopy Systems
Laser scanning confocal microscopy is one of the leading technologies in advanced biological imaging. Based on the principle of rejecting out-of-focus light using a confocal pinhole, it enables the acquisition of fine optical sections within thick samples, allowing precise three-dimensional reconstructions.
Initially designed as an improvement over conventional fluorescence microscopy, it has gradually evolved into an integrated platform combining high-precision optics, advanced photonics, and computational analysis.
Introducto to the confoncal Microscopy
Principles of Confocal Microscopy
This article highlights the transformative impact and recent advancements of confocal laser scanning microscopy in biological research, showcasing its pivotal role in enhancing cellular and molecular imaging.
Architecture of Modern Confocal Systems
Contemporary confocal systems rely on an optimized technological architecture, including:

Optical System
High numerical aperture apochromatic objectives
Advanced chromatic aberration correction
Enhanced mechanical and thermal stability

Excitation Sources
Stabilized multi-wavelength lasers
Rapid laser line switching
Precise power control to reduce phototoxicity

Software Environment
Automated interfaces
Programmable protocols
AI-assisted analysis

Spectral Control
Full spectral acquisition (lambda imaging)
Separation of overlapping fluorophores
Integrated spectral unmixing
This review highlights the recent advancements, versatile applications, and ongoing challenges of Confocal Laser Scanning Microscopy (CLSM), showcasing its pivotal role in bridging conventional and high-resolution imaging across fields from medicine to materials science."
Major Technological Advances (Last Two Decades)
Over the past twenty years, confocal microscopy has evolved toward faster, more sensitive, and more analytically powerful systems.
Improved Sensitivity and Photon Efficiency
Increased quantum efficiency
Reduced photobleaching
Lower phototoxicity
This allows prolonged imaging of live samples.
Adaptive Optics and Deep Tissue Imaging
Recent systems include:
Dynamic aberration correction
Enhanced deep-tissue contrast
Improved penetration in thick tissues
Super-Resolution Integration
Some platforms incorporate:
Multi-element detection
Advanced computational deconvolution
Confocal–super-resolution hybrid techniques
These approaches improve lateral resolution while maintaining the flexibility of classic confocal microscopy.
High-Speed and Dynamic Imaging
Modern systems integrate:
Resonant scanning
Spinning disk technology
Rapid volumetric acquisition
They enable:
High-frame-rate 3D imaging
4D reconstruction (time + volume)
Study of fast biological processes (calcium signaling, intracellular trafficking)
Artificial Intelligence and Quantitative Analysis
AI is transforming experimental workflows through:
Intelligent denoising
Automated segmentation
3D morphometric quantification
Colocalization analysis
Extraction of multiparametric metrics
Confocal microscopy thus becomes a reproducible quantitative analysis tool.
These articles highlight the latest advancements in confocal laser scanning microscopy, from enhanced sensitivity and photon efficiency using deep learning, to high-speed dynamic imaging systems, and the integration of super-resolution techniques that push the boundaries of optical microscopy
📄Research on Reflective High‑Speed Multi‑Point Confocal Microscopy System
(H. Hu et al., 2025)"This article showcases recent innovations in high‑speed confocal imaging, presenting a reflective multi‑point system that advances dynamic observation capabilities for fast biological processes."
"This article highlights cutting‑edge improvements in confocal microscopy sensitivity and photon efficiency, demonstrating how deep learning algorithms can enhance real‑time resolution beyond conventional limits."
📄 The Development of Microscopy for Super‑Resolution
(Sheppard, 2021)"This review explores the evolution of microscopy toward super‑resolution, including developments in confocal techniques, and highlights how these advancements are redefining imaging performance."

Modern Confocal Platforms: Modularity and Integration
Current platforms are designed as modular, configurable systems. They allow integration with:
Multiphoton imaging
Super-resolution
Light-sheet microscopy
FLIM (fluorescence lifetime imaging)
Quantitative FRET
They combine:
Precision optics
Advanced photonics
Scientific computing
Experimental automation
Applications of Modern Confocal Systems
Contemporary confocal microscopes are particularly suited for:

Fundamental Research
High-resolution 3D/4D imaging
Dynamic live-cell studies
Quantitative colocalization analysis

Neuroscience
Fast calcium imaging
Synaptic mapping
Study of neuronal networks

Translational and Clinical Research
Confocal endomicroscopy
Optical dermatology
Gastroenterological applications

Advanced Cell Biology
3D organoids
Tumor spheroids
Microenvironment analysis
Overall, modern confocal microscopy serves as a versatile and indispensable imaging platform, enabling researchers to visualize complex biological structures and dynamic processes with unprecedented spatial and temporal resolution. Its broad applicability from fundamental cell biology to neuroscience, organoid research, and clinical imaging underscores its central role in advancing both basic science and translational biomedical studies.

Theory of Confocal Microscopy
Fundamental Principles
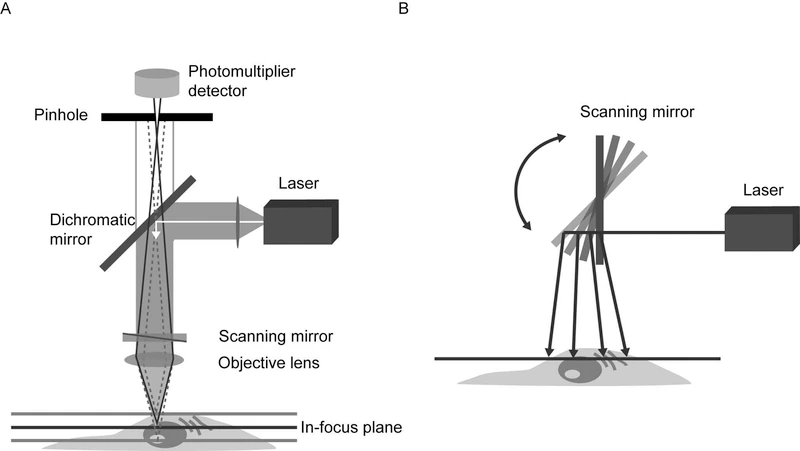
Confocal microscopy is based on the principle of spatial filtering, which eliminates out-of-focus light to obtain fine optical sections of a three-dimensional sample.
Unlike wide-field microscopy, where the entire sample thickness is illuminated simultaneously, confocal microscopy:
Illuminates a precise point of the specimen using a focused laser beam
Uses a confocal pinhole placed in a plane conjugate to the focal plane
Rejects light originating from planes above and below the focus
The final image is reconstructed point by point through controlled beam scanning.
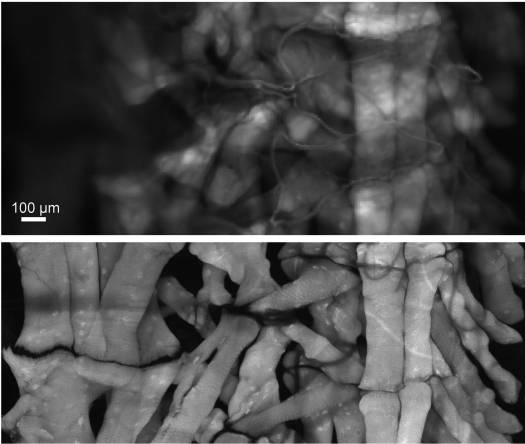
Figure : Widefield vs Confocal Microscopy: Enhanced Resolution and Optical Sectioning in Drosophila Muscle Imaging"
Comparison of widefield and confocal microscopy of a Drosophila larval fillet hemisegment stained with AlexaFluor 647‑phalloidin to label muscle. The widefield image was collected in 1 second, while the confocal microscopy image (pinhole = 1 Airy Unit) using a 20x objective required ~2 hours, demonstrating confocal microscopy’s superior optical sectioning and resolution."
Image Formation and Point Spread Function (PSF)
Resolution in confocal microscopy is determined by:
The numerical aperture (NA) of the objective
The excitation and emission wavelengths
The pinhole diameter
The optical response of the system is described by the Point Spread Function (PSF).
Confocal microscopy improves axial resolution compared to wide-field microscopy by reducing the detection volume.
Typical modern resolution:
Lateral: ~180–220 nm
Axial: ~500–700 nm
Optimized systems combined with deconvolution or Airyscan modules can achieve even higher performance.
Fluorescence Excitation and Emission
Fluorescence is the primary imaging mode in confocal microscopy. The process involves:
Absorption of an excitation photon
Transition to an excited state
Return to the ground state with emission of a longer-wavelength photon
Critical parameters include:
Absorption and emission spectra
Quantum yield
Fluorescence lifetime
Photostability
Modern systems employ spectral unmixing to separate fluorophores with overlapping spectra.
Signal-to-Noise Ratio and Sensitivity
The measured signal is proportional to the number of detected photons, but is influenced by:
Photon noise (shot noise)
Detector electronic noise
Sample autofluorescence
Hybrid detectors (HyD) and GaAsP detectors significantly improve sensitivity and signal-to-noise ratio.
Modern AI-based denoising algorithms enhance image quality without increasing phototoxicity.

Adaptive Optics
Recent systems incorporate adaptive optics to correct aberrations induced by thick tissues.
This enables:
Better deep-tissue penetration
Improved resolution in live tissues
More stable intravital imaging
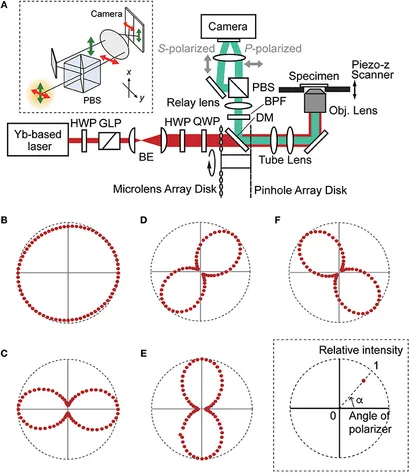
Figure: Optical Configuration and Polarization Control of the TPM-SD System
(A) Schematic of the developed two-photon microscopy–spinning disk (TPM-SD) system, illustrating key components: beam expander (BE), bandpass filter (BPF), dichroic mirror (DM), Glan-Laser polarizer (GLP), half-wave plate (HWP), polarizing beam splitter cube (PBS), and quarter-wave plate (QWP). The upper inset depicts the polarization-resolving detection setup. (B–F) Controlled polarization states of the incident light at the specimen achieved by adjusting HWP and QWP: (B) circular polarization (ellipticity 0.95); (C–F) linear polarization at 0° (ellipticity 0.28), 45° (0.27), 90° (0.27), and 135° (0.24). The lower inset illustrates the method for plotting relative intensities on a polar coordinate system.
Theoretical Limits and Diffraction Breaking
The classical diffraction limit (Rayleigh criterion) constrains resolution.
Modern extensions include:
Integrated STED confocal
Airyscan detection
Advanced computational deconvolution
Confocal–super-resolution hybrid techniques
These approaches allow surpassing the ~200 nm diffraction limit under optimized conditions.
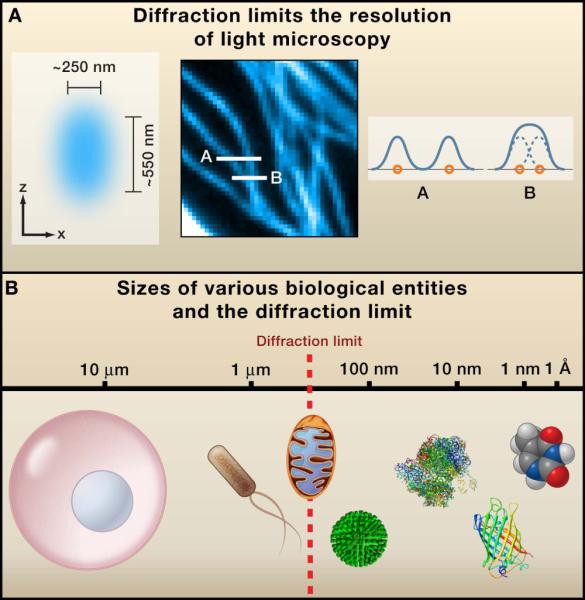
Figure: Diffraction-Limited Resolution in Conventional Light Microscopy
(A) The focal spot of a high numerical aperture objective, illustrated as a cyan ellipsoid, exhibits lateral and axial widths of ~250 nm and ~550 nm, respectively. The point spread function (PSF) of a point emitter imaged through the objective displays similar dimensions, defining the diffraction-limited resolution. Two objects separated by distances larger than this limit are resolvable, while closer objects appear as a single entity. This is demonstrated by the cross sections of a microtubule image (cyan curves A and B) corresponding to positions indicated by white lines in the middle panel.
(B) Comparative size scale of biological structures relative to diffraction-limited resolution, including a mammalian cell, bacterial cell, mitochondrion, influenza virus, ribosome, green fluorescent protein, and small molecule (thymine).
Modern Modeling and Quantification
Current platforms integrate:
Automated colocalization quantification
3D morphometric analysis
Deep learning–based cell segmentation
Multi-parametric dynamic analysis
Modern confocal microscopy is no longer purely descriptive it has become an advanced quantitative tool.
Conclusion
Modern confocal microscopy has evolved far beyond its original role as a high-resolution imaging tool. Today, it represents a fully integrated platform combining precision optics, multi-wavelength photonics, advanced photon detection, computational analysis, and artificial intelligence. These systems enable quantitative, dynamic, and high-dimensional imaging across a wide range of biological and biomedical applicationsfrom live-cell imaging and organoid studies to neuroscience research and translational clinical investigations. By bridging classical optical techniques with cutting-edge computational methods, confocal microscopy has become an indispensable resource for researchers and clinicians seeking to uncover the intricate details of life at the cellular and tissue levels.