Dynamic Live‑Cell Imaging with Photobleaching:
FRAP and FLIP in Modern Cell Biology
Fluorescence photobleaching techniques especially Fluorescence Recovery After Photobleaching (FRAP) and Fluorescence Loss In Photobleaching (FLIP) are foundational methods used to measure the mobility, diffusion, turnover, and connectivity of biomolecules in living cells. With the advent of genetically encoded fluorescent proteins (like GFP and its derivatives), these methods enable highly quantitative, non‑invasive investigations of intracellular dynamics in real time.
What is FRAP?
FRAP is a live-cell imaging method that measures the mobility and turnover of fluorescently labeled molecules. By selectively photobleaching a region of interest and tracking the fluorescence recovery, FRAP provides insights into the diffusion dynamics, binding interactions, and intracellular trafficking of proteins, lipids, and other macromolecules.
Principle of FRAP
A targeted region in a fluorescently labeled cell or tissue is exposed to high-intensity light, irreversibly bleaching the fluorophores.
Fluorescence recovery in the bleached area is monitored over time.
The rate and extent of recovery reflect the mobility and exchange of molecules within the local environment.
FRAP allows researchers to distinguish between freely diffusing molecules, transiently bound molecules, and immobile fractions, giving quantitative insight into molecular behavior in membranes, organelles, or cytoplasmic compartments.
Parameters Measured by FRAP
FRAP experiments generate quantitative metrics such as:
Diffusion coefficient (D): Indicates the speed of molecular movement.
Mobile fraction: Proportion of molecules able to diffuse freely.
Immobile fraction: Molecules stably associated with structures.
Turnover rate: Dynamics of molecular exchange in specific subcellular regions.
These measurements are fundamental for understanding membrane fluidity, cytoskeletal dynamics, protein trafficking, and molecular interactions.
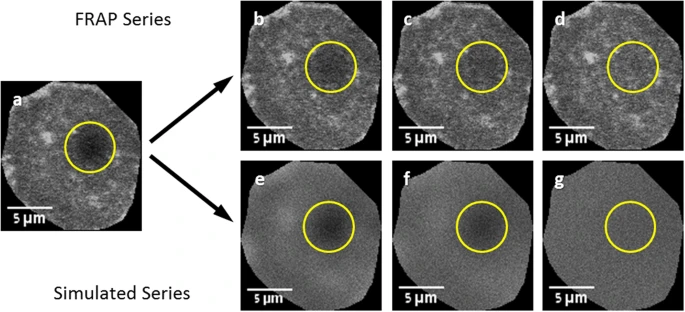
Figure: Fluorescence Recovery After Photobleaching (FRAP) Experimental and Simulated Recovery
This figure illustrates a FRAP experiment alongside its corresponding simulated recovery series. (A) The first post-bleach frame serves as the reference for generating the simulated recovery. (B–D) Selected frames from the actual FRAP series show gradual restoration of fluorescence within the bleached spot over time. (E–G) Corresponding frames from the simulated recovery series depict the predicted fluorescence restoration, allowing comparison with experimental data. In all panels, the circle indicates the user-defined region used to monitor fluorescence recovery, highlighting the dynamics of molecular diffusion and protein mobility within the bleached area.
Modern Applications of FRAP
Membrane and Organelle Dynamics
FRAP remains crucial for measuring protein and lipid diffusion in plasma membranes, endoplasmic reticulum, Golgi apparatus, and nuclear membranes. Differences in recovery curves reveal localized constraints, compartmentalization, or tethering to structural scaffolds.
Cytoskeletal Turnover
By labeling cytoskeletal proteins with fluorescent tags, FRAP can track actin filaments, microtubules, and intermediate filaments to determine whether turnover occurs at filament ends or along the filament body. This enables precise mapping of cytoskeletal remodeling in response to signaling, mechanical stress, or drug treatments.
Computational and High-Throughput FRAP
Recent advances integrate machine learning and automated image analysis to fit FRAP recovery curves using diffusion-reaction models, allowing high-throughput quantification of molecular mobility under multiple conditions.
What is FLIP?
FLIP is a complementary technique that measures fluorescence loss due to repeated photobleaching at a defined site. By continuously bleaching a small region and monitoring fluorescence in the rest of the cell, FLIP evaluates molecular connectivity, compartmental continuity, and exchange pathways.
Principle of FLIP
A specific area is repeatedly photobleached.
Molecules that diffuse freely throughout the cell cause progressive fluorescence loss in non-bleached regions.
The kinetics of fluorescence decay reveal the degree of connectivity and barriers to molecular movement.
FLIP is particularly effective for studying membrane networks, organelle continuity, and intracellular molecular trafficking.
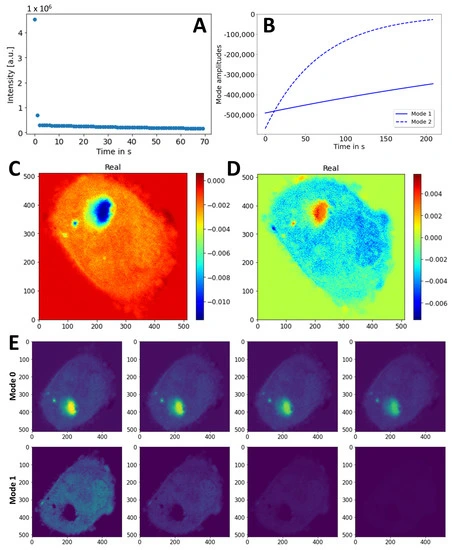
Figure: Dynamic Mode Decomposition of FLIP Image Sequences in CHO Cells Expressing eGFP-Q145
This figure presents the analysis of eGFP-Q145 dynamics in CHO cells using Dynamic Mode Decomposition (DMD) of a FLIP (Fluorescence Loss in Photobleaching) image sequence. (A) Singular values calculated from the reshaped image stack provide insight into the dominant dynamic components of the dataset. (B) Mode decays extracted by DMD using rank 2 show temporal evolution of the two primary dynamic modes. (C, D) Two-dimensional spatial maps of the real parts of the identified dynamic modes are displayed: mode 0 (C) and mode 1 (D), while imaginary components were negligible and are omitted. (E) Reconstruction of the dynamics for mode 0 (upper panel) and mode 1 (lower panel) with identical intensity scaling; every 20th frame is shown to illustrate progression. This analysis highlights the ability of DMD to decompose complex intracellular fluorescence dynamics into interpretable spatial and temporal modes.
Biological Insights from FRAP and FLIP
Membrane Continuity: Both methods can confirm continuity in the endoplasmic reticulum, Golgi apparatus, and nuclear membranes, demonstrating unrestricted lateral diffusion of proteins and lipids.
Intracellular Barriers: FLIP identifies restricted diffusion zones, such as specialized neuronal domains or organelle boundaries.
Molecular Turnover: FRAP allows quantification of protein lifetimes and exchange rates in organelles, cytoskeletal structures, and membraneless compartments.
Phase Separation Dynamics: FRAP is increasingly used to study biomolecular condensates, providing insights into fluid versus gel-like properties of intracellular compartments.
Advantages of Modern FRAP and FLIP
Non-invasive and real-time imaging of living cells.
Compatibility with genetically encoded fluorescent proteins like GFP, mCherry, or mScarlet.
Integration with high-resolution confocal and super-resolution microscopy.
Enhanced quantitative analysis through computational modeling, allowing precise extraction of diffusion coefficients, binding kinetics, and mobile/immobile fractions.
Best Practices for FRAP and FLIP Experiments
To ensure reliable and interpretable results:
Include non-bleached control regions for normalization.
Correct for general photobleaching caused by imaging.
Select fluorescent proteins with high photostability.
Optimize laser intensity and imaging frequency for reproducibility.
Apply mechanistic models for recovery or loss curves to extract biologically meaningful parameters.
Conclusion
FRAP and FLIP remain cornerstone techniques in modern cell biology, biophysics, and molecular research. By enabling the quantitative measurement of molecular mobility, turnover, and connectivity, they provide unparalleled insights into intracellular dynamics, membrane fluidity, cytoskeletal remodeling, and biomolecular compartmentalization. Advances in fluorescent protein technology, imaging hardware, and computational modeling have expanded their utility, making them essential tools for investigating complex cellular processes in health and disease.