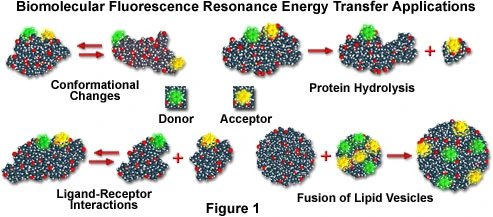
Here is an optimized and more concise scientific version of your text. I kept the same information but improved clarity, flow, and academic tone, and removed repetition.
Fluorescent and Bioluminescent Proteins
Many living organisms are capable of emitting light through biochemical mechanisms, including fireflies, jellyfish, and certain bacteria. In some organisms, light emission occurs through the absorption of specific wavelengths of light, while in others it results from chemical reactions that release stored energy.
Fluorescent proteins emit light by absorbing energy from an external light source and re-emitting it at a longer wavelength. Such proteins are found in organisms such as jellyfish, coral, sea anemones, and certain bacteria. In contrast, bioluminescence results from an internal chemical reaction. Examples include the firefly and species of the Noctiluca genus. In these organisms, the light-emitting molecules may be located inside the cells (as in fireflies and Noctiluca) or secreted outside the body (as in Cypridinacea). Although proteins participate in these reactions, they do not directly emit light themselves.
Both fluorescent proteins and bioluminescent systems are widely used in medical and biological research. Although only small quantities of these proteins were initially isolated from natural sources, they are now produced through chemical synthesis or genetic engineering. The genes encoding Green Fluorescent Protein (GFP) and its variants are commercially available and widely applied in cell biology and molecular biology for visualizing cellular processes.
Fluorescent Proteins
Important groups of fluorescent proteins include:
Green fluorescent protein (GFP) and its variants: BFP, CFP, GFP, YFP, and DsRedFP
Phycobiliproteins: B-Phycoerythrin (B-PE), R-Phycoerythrin (R-PE), and Allophycocyanin (APC)
GFP naturally occurs in jellyfish. Fluorescence originates from specific amino acids located at positions 65–67 of the protein, which emit light when excited by ultraviolet or blue light. DsRed, another fluorescent protein, is found in sea anemones. The GFP gene is widely used as a reporter gene to visualize and track cellular activity through its expression inside living cells.
Phycobiliproteins are derived from bacteria and algae and contain covalently linked fluorescent groups. Specimens labeled with these proteins are commonly used in flow cytometry and can also be observed using fluorescence microscopy.
Excitation and Emission Characteristics of Fluorescent Proteins
| Fluorescent Protein | Excitation Wavelength (nm) | Emission Wavelength (nm) |
|---|---|---|
| GFPwt (Wild Type) | 395 / 475 | 510 |
| ECFP | 433 | 475 |
| EGFP | 488 | 507 |
| EYFP | 513 | 527 |
| DsRed | 558 | 583 |
| B-PE | 545 / 565 | 575 |
| R-PE | 410 / 545 / 565 | 578 |
| APC | 650 | 660 |
Table 1: Excitation and emission properties of selected fluorescent proteins.
Bioluminescent Proteins
Two of the most commonly used bioluminescent systems in biological research are aequorin and the luciferin–luciferase system.
Aequorin is a bioluminescent protein complex isolated from jellyfish and widely used to measure intracellular calcium ion concentrations. It consists of the protein apoaequorin and a luminescent molecule. When calcium ions bind to aequorin, the complex dissociates and emits blue light at approximately 466 nm. Unlike fluorescent dye probes, measurements using aequorin do not require excitation light and are therefore not affected by autofluorescence. Synthetic apoaequorin and its gene are available and can be expressed in cellular compartments such as mitochondria.
Luciferin is a light-emitting molecule found in fireflies. When oxidized by the enzyme luciferase, it produces luminescent light with wavelengths up to approximately 560 nm. Because ATP is required for the luciferin–luciferase reaction, this system is widely used in highly sensitive ATP detection assays. Commercial ATP detection kits based on this reaction are commonly used in research and biotechnology. The luciferase gene can also be expressed inside cells as a reporter system.