Specimen Preparation Using Synthetic Fluorophores and Immunofluorescence
Target: Intermediate Filament Stains (e.g., vimentin, desmin)
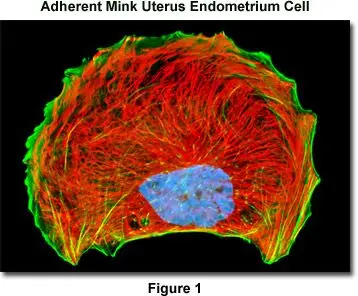
This protocol outlines immunofluorescent staining of adherent cells for intermediate filaments, actin filaments, and nuclei using primary antibodies and low-molecular-weight synthetic fluorophores such as Texas Red-X, Alexa Fluor, rhodamine, fluorescein, and cyanine dyes.
1. Reagents Preparation
Cytoskeletal Buffer (CB)
60 mM PIPES, 27 mM HEPES, 10 mM EGTA, 4 mM MgSO₄·7H₂O in 1 L water
Adjust pH to 7.0
Phosphate Buffered Saline (PBS) with Ca²⁺ & Mg²⁺
KCl 0.2 g, KH₂PO₄ 0.2 g, NaCl 8 g, Na₂HPO₄·7H₂O 1.74 g
Add CaCl₂·2H₂O 0.132 g and MgCl₂·6H₂O 0.10 g
Adjust pH to 7.2
Mixed Aldehyde & Detergent Fixative (fresh daily)
3% paraformaldehyde in CB + 0.3% Triton X-100 + 0.05% glutaraldehyde
Blocking Buffer
10% normal goat serum (NGS) in PBS with 0.05% Triton X-100 + 2–3 mg sodium azide/100 mL
PBS-Triton Wash Buffers
Simple wash: PBS + 0.05% Triton X-100
Wash with blocking serum: PBS + 0.05% Triton X-100 + 1% NGS
Primary Antibody Cocktail
Primary antibodies in 50% Blocking Buffer + PBS-Triton Wash Buffer (final 5% NGS)
Secondary Antibody / Phalloidin Cocktail
Secondary antibody + fluorophore in 50% Blocking Buffer + PBS-Triton Wash Buffer (final 5% NGS)
Optional: phalloidin conjugates added for actin staining
Nuclear Stains
DAPI: 5 μL of 10 mg/mL stock in 150 mL 50% PBS for 5 min
Hoechst 33342/33258: 5 μL of 10 mg/mL stock in 150 mL Hanks BSS for 30 min
SYTOX Green/Orange: 10 μL of 5 mM stock in 250 mL Hanks BSS for 30 min
Cyanine Dyes / DRAQ5: Dilute 1:20–1:1000 in PBS, treat 5–30 min
2. Cell Preparation & Fixation
Aspirate medium from Petri dishes with adherent cells.
Wash cells twice with pre-warmed CB buffer (37°C, 5 min each).
Fix cells with mixed aldehyde fixative (37°C, 10 min).
Wash once with CB, then twice with PBS-Triton Wash Buffer (5 min each).
3. Blocking
Incubate cells in 10% NGS Blocking Buffer for 60 min at room temperature on an orbital shaker (5–10 rpm).
4. Primary Antibody Staining
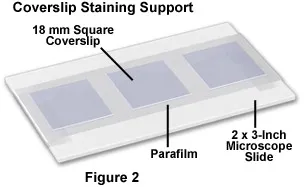
Prepare Parafilm-covered slides for coverslip staining.
Place coverslips cell-side down on 100 μL drops of primary antibody cocktail.
Incubate in a humidity chamber at 37°C for 1.5 hours.
Wash three times with PBS-Triton Wash Buffer + Blocking Serum (5–10 min each, orbital shaker).
5. Secondary Antibody / Phalloidin Staining
Place coverslips on 100 μL drops of secondary antibody/phalloidin cocktail.
Incubate in humidity chamber at 37°C:
1 hr for smaller antibody fragments
1.5 hr for full IgG molecules
Cover chamber with aluminum foil to protect fluorophores.
Wash three times with PBS-Triton Wash Buffer + Blocking Serum (5–10 min each).
6. Nuclear Counterstaining
Wash cells twice with PBS-Triton Wash Buffer.
Apply diluted nuclear dye:
DAPI / cyanine dyes: 5–30 min, protect from light
Hoechst / SYTOX: 30 min in Hanks BSS
Wash three times with PBS or Hanks BSS (5 min each).
Optional: wash 2–3 times with distilled water if air-drying coverslips overnight.
7. Mounting
Carefully remove coverslips, wipe excess water.
Lean coverslips cell-side down against a labeled Petri dish lid; air-dry overnight, protected from light.
Mount coverslips on clean microscope slides using appropriate mounting medium.

Notes
Maintain consistent temperature and light protection to prevent photobleaching.
Test antibody compatibility if combining multiple primary antibodies.
Ensure distilled water washes before air-drying to avoid salt crystals on the coverslip.