Collisional Quenching and Cytoskeletal Visualization in COS-7 Cells
A common phenomenon in fluorescence microscopy is collisional quenching, which occurs when an excited-state fluorophore interacts with a non-fluorescent molecule in solution, resulting in deactivation of the fluorophore and a return to the ground state. Typically, neither molecule undergoes a permanent chemical change. Various simple elements and compounds including oxygen, halogens, amines, and electron-deficient organic molecules can act as collisional quenchers. This process can provide insights into the local molecular environment, as quenchers diffuse or undergo conformational changes that allow them to collide with fluorophores during their excited-state lifetime. Mechanisms of collisional quenching include electron transfer, spin-orbit coupling, and intersystem crossing to the excited triplet state. Terms such as dynamic quenching or internal conversion are often used interchangeably with collisional quenching.
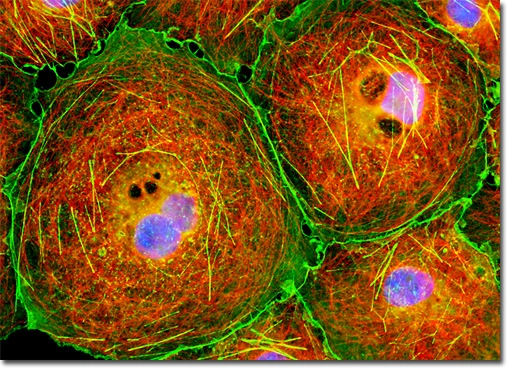
In the image presented, an adherent culture of COS-7 African green monkey kidney fibroblast cells was immunofluorescently labeled to visualize cytoskeletal and nuclear structures. Microtubules were targeted using primary anti-tubulin mouse monoclonal antibodies followed by goat anti-mouse Fab fragments conjugated to Cy3. Simultaneously, the filamentous actin network was stained with Alexa Fluor 488-conjugated phalloidin, and nuclei were labeled with DAPI. Images were acquired in grayscale using a QImaging Retiga Fast-EXi camera mounted on an Olympus BX-51 microscope equipped with bandpass emission fluorescence filter blocks from Omega Optical. During post-processing, individual image channels were pseudocolored using RGB values corresponding to the emission spectra of each fluorophore, providing a clear and visually distinct representation of microtubules, actin filaments, and nuclei.
This dual- and triple-labeling approach allows detailed visualization of the cytoskeletal architecture in relation to nuclear organization, providing valuable insights into cellular morphology, spatial interactions between cytoskeletal components, and potential changes under experimental conditions.