Resolution and Contrast in Confocal Microscopy
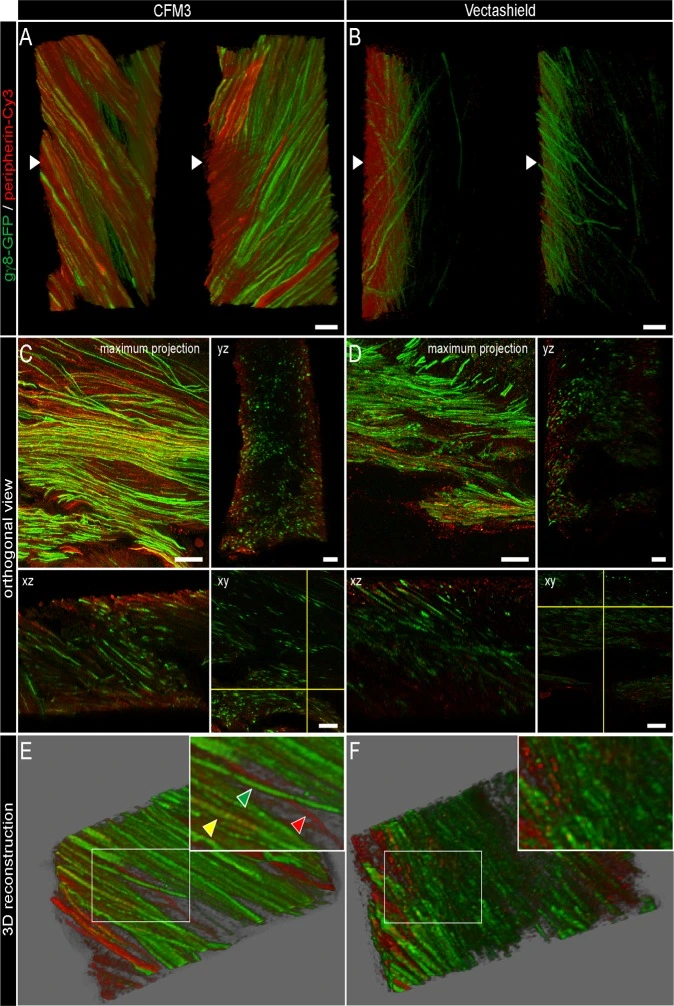
Figure: Improvement of axial resolution in fixed mouse olfactory bulbs
Fifty-micron vibratome sections of olfactory bulb from transgenic mice were processed for double immunofluorescence detection of Green Fluorescent Protein (GFP, green) and peripherin (red), and mounted in CFM3 or Vectashield. Confocal datasets were deconvolved and analyzed in 3D.
Sections mounted in CFM3 show superior axial resolution and signal preservation, enabling clear discrimination of three axonal subpopulations: GFP+/peripherin− (green), GFP−/peripherin+ (red), and double-labeled GFP+/peripherin+ (yellow). In contrast, sections mounted in Vectashield exhibit reduced contrast and diminished capacity to distinguish these labeled axon categories. Scale bars: 10 μm.
Understanding Resolution Limits
All optical microscopes, including widefield, confocal, and two-photon systems, are constrained by fundamental physical limits. In an ideal system, resolution depends on the numerical aperture (NA) of the objective and the wavelength of light used for excitation and emission. Resolution is inherently linked to contrast, defined as the minimum separation between two points at which they remain distinguishable. In fluorescence microscopy, contrast is affected by photon collection efficiency, detector dynamic range, optical aberrations, and pixel density. Noise further reduces the visibility of closely spaced features, emphasizing the practical interplay between resolution, contrast, and imaging conditions.
Point Spread Function and the Airy Disk
A point-like light source produces a point spread function (PSF) that characterizes how the microscope blurs a single point in space. In lateral dimensions, this is represented by the Airy disk, a diffraction pattern with a central bright spot surrounded by concentric rings. The separation of two point sources is directly related to the PSF width, often quantified as the full width at half maximum (FWHM). Confocal microscopy sharpens this PSF by combining point illumination and point detection, reducing lateral and axial spread by roughly 30% compared to widefield imaging.
Lateral and Axial Resolution
Lateral resolution ((r_\text{lateral})) in confocal systems is approximately:
[r_\text{lateral} = 0.4 \frac{\lambda}{NA}]
where (\lambda) is the emission wavelength and NA is the objective numerical aperture. Axial resolution ((r_\text{axial})) depends more strongly on NA and refractive index ((n)):
[r_\text{axial} = 1.4 \frac{\lambda n}{NA^2}]
Confocal microscopes offer modest axial improvement over widefield systems, but their true strength lies in optical sectioning, allowing three-dimensional imaging of thick, inhomogeneous specimens.
Practical Factors Affecting Contrast
Real-world contrast is influenced by photon statistics, optical aberrations, and the digital sampling process. Confocal images are recorded as discrete pixels, introducing potential contrast loss if the pixel size does not match the resel (resolution element). The Nyquist criterion ensures accurate sampling by requiring at least two pixels per smallest resolvable feature, avoiding undersampling artifacts such as aliasing. Oversampling can improve localization of features but increases photobleaching risk.
Optimizing Imaging Conditions
Optimal confocal imaging balances optical resolution, sampling frequency, and detector sensitivity. Adjusting pinhole size, NA, illumination intensity, and zoom settings allows control over PSF dimensions and image contrast. In some cases, intentionally reducing resolution (larger pinholes or lower NA) can prevent oversampling and minimize photodamage while maintaining optical sectioning.
Conclusion
Resolution in confocal microscopy is not a fixed value but a function of contrast, sampling, and experimental conditions. Understanding the interplay between optical physics, digital sampling, and specimen limitations enables microscopists to achieve the best practical imaging performance while preserving specimen integrity.